As the COVID-19 case count and the death toll from the virus continues to surge in California, plans for the reopening of in-person classes at Stanford’s Graduate School of Business have become increasingly complicated and stubbornly challenging. Shifting state and county guidelines, still-to-be-unveiled university protocols along with the spread of […]
Is it safe to go back to the gym? Here’s what you need to know — Recommendations are independently chosen by Reviewed’s editors. Purchases you make through our links may earn us a commission. After months of being closed, gyms and fitness studios have started to reopen in 43 states amid the […]
We’re keeping track of the latest news regarding the coronavirus in South Florida and around the state. Check back for updates throughout the day. COVID-19 hits two of Miami-Dade’s largest police departments as enforcement ramps up 1:15 p.m.: The rapid recent rise in coronavirus infections in South Florida has begun […]
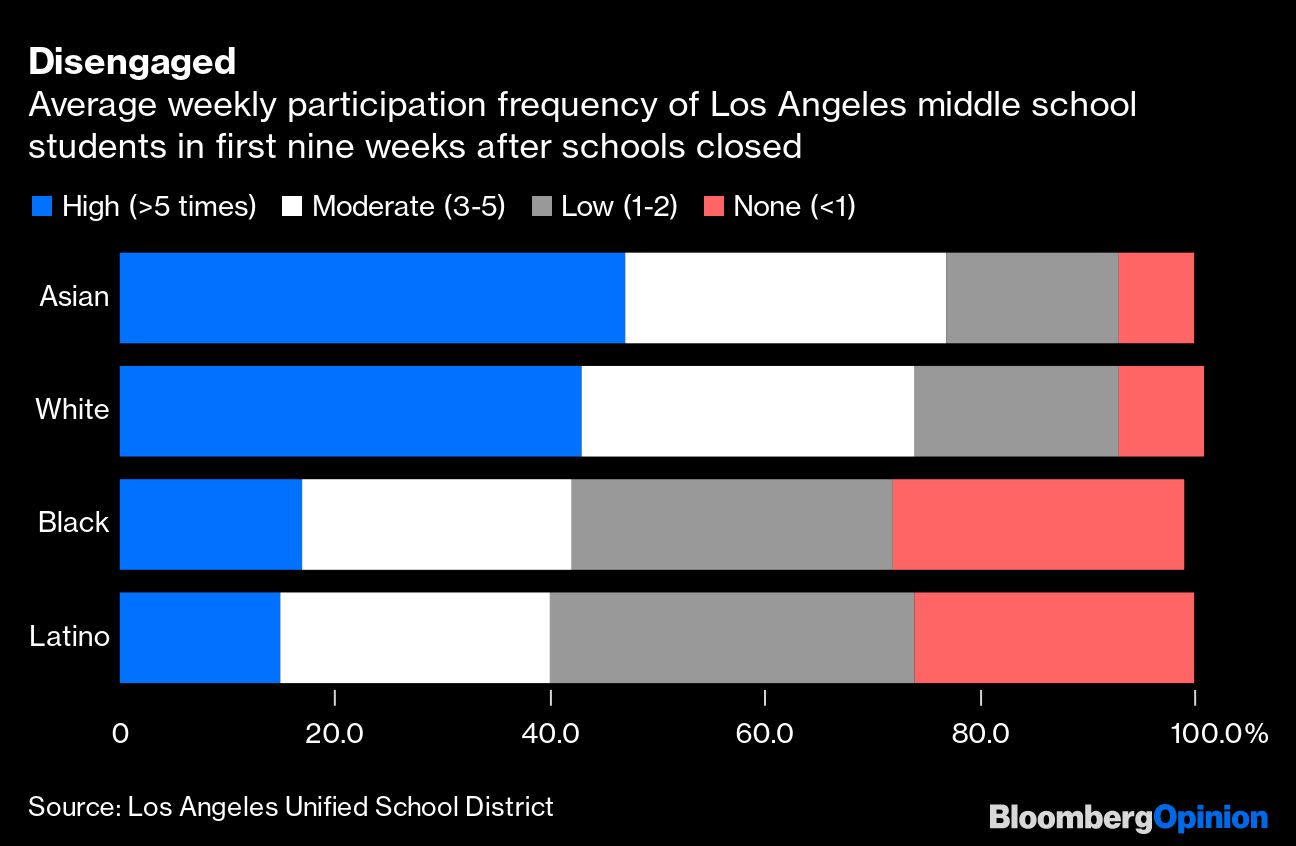
(Bloomberg Opinion) — When the Los Angeles Unified School District announced on Monday that it will not resume any in-person instruction this fall, it was a political victory for teachers and a defeat for families, science and opportunity for all. The teachers’ union opposed reopening schools amid the continuing rise […]
While the outbreak of COVID-19 has crumbled several businesses, many companies in the consumer staples space appear to be on firm grounds. One such consumer staple player benefiting from such trends is Helen of Troy Limited HELE, with its shares up as much as 40.6% in the past three months […]
Click here to read the full article. Comfort customers are playing hard to get these days. As the ongoing pandemic has spurred many to add a morning walk or run to their daily regimens, they’re heading to comfort stores looking for a pair of performance sneakers over casuals. Brands such […]
It was another troubling week in California, as growing infections and hospitalizations prompted state and local officials to announce new widespread closures of businesses and schools across the state to slow the coronavirus pandemic. The pandemic, which is disproportionately ravaging communities of color, has killed at least 7,345 Californians. This […]
At the end of June, with many of the state’s key coronavirus metrics trending in a positive direction, it appeared as if Hampton Roads had escaped the worst of the pandemic. And with those promising signs came the hope that schools could reopen this fall — as close to normally […]