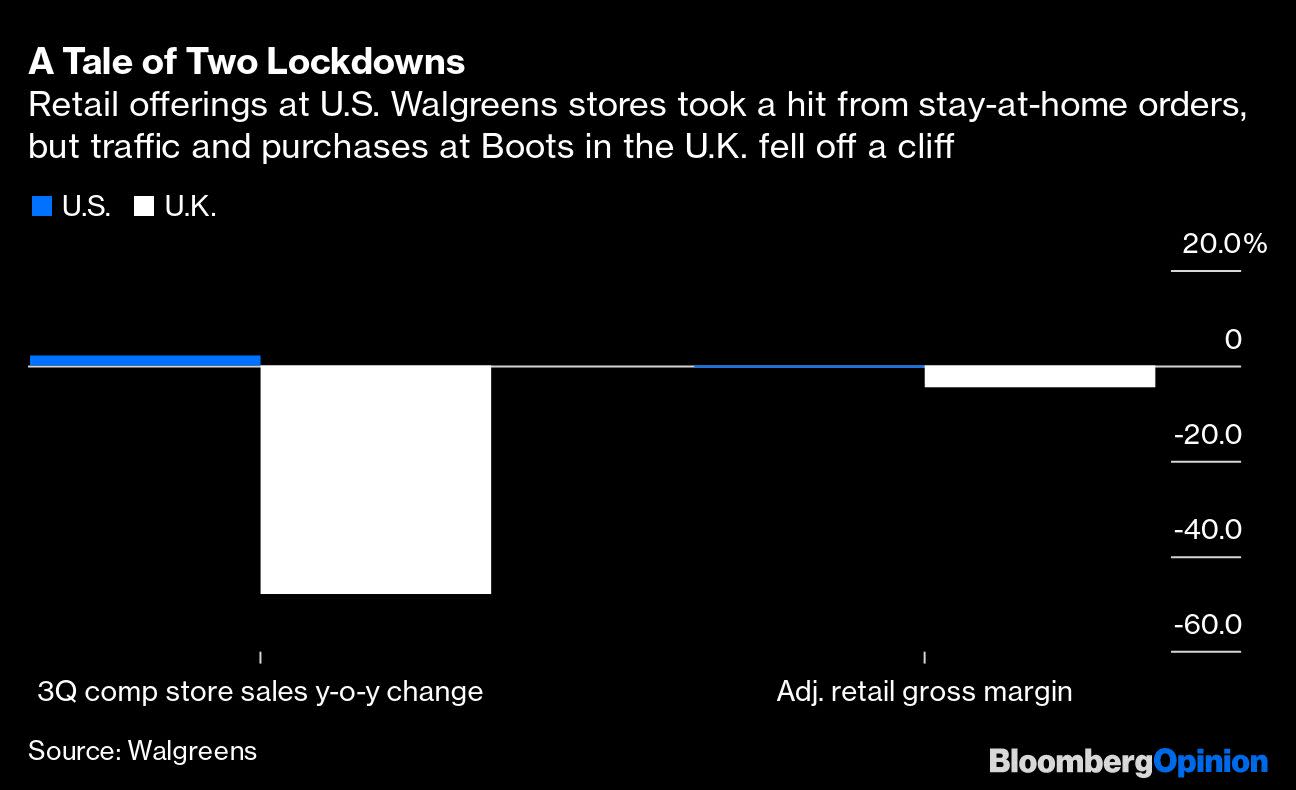
(Bloomberg Opinion) — Health-care earnings season is firmly here, with reports now in from three pillars of the sector: pharmacy giant Walgreen Boots Alliance Inc., health insurer UnitedHealth Group Inc. and drug-and-device conglomerate Johnson & Johnson. Against the backdrop of a global pandemic, each had a singular quarter. And while experiences varied given the different niches […]
COVID
The novel coronavirus pandemic is keeping Americans away from the doctor’s office. For most people, that means little more than postponing a dental checkup or enduring a minor illness at home. But those with chronic medical conditions ― especially ailments that make them more susceptible to infections like COVID-19 ― […]
AerialCLT, a Charlotte studio known for its aerial silks and trapeze classes, is teetering on the potential of a permanent closure due to the COVID-19 crisis — but the owner isn’t giving up. In an Instagram announcement on Tuesday, the studio wrote: “AerialCLT Family, we have had 8.5 years of […]
NEWARK, NJ — Recovering from the coronavirus shutdown has been a slow process, Newark. But there’s good news: the “new normal” is getting closer. Newark’s restaurants, stores, businesses and other cultural institutions continue to reawaken from the COVID-19 shutdown that threw a devastating monkey wrench into New Jersey’s economy. A […]
The worst pandemic in modern memory has inspired a massive effort to harness intellectual property rights. More than 1,500 trademark applications have been filed with the U.S. Patent and Trademark Office for variations on the COVID-19 and coronavirus theme. They range from the serious to the silly, from the innovative […]
A basketball objective and two water slides full the pool facilities. If updated art has sometimes been related to abstract painting, it can not in any means be decreased to this affiliation. Figurative art is making a comeback on the present art work scene and artists are using the kind […]
When President Trump promoted the malaria drug hydroxychloroquine as a possible treatment for COVID-19 in the spring, demand for the product jumped. Most of the subsequent debate has been about the efficacy of the drug as a treatment for prophylaxis for COVID-19. But arguably as important is what happens to […]
Image caption The pharmacy in John Wood Street is one of two businesses linked to the cluster A link has been established between a cluster of Covid-19 cases in Inverclyde and an earlier outbreak at a call centre in Lanarkshire. NHS Greater Glasgow said 13 people had tested positive in […]